Medical Device Design History File Template
Here are some of the images for Medical Device Design History File Template that we found in our website database.
MEDICAL EQUIPMENT Wallpapers Wallpaper Cave

Program Spotlight: Biomedical Equipment Technology DCTC News

10 things you didn t know about medical residents AAMC

Medical Laboratory Technology Mississippi Gulf Coast Community College

Medical Diagnosis

Medical Laboratory Technology Uwi at Margaret Hensley blog

HD Medical Wallpapers Top Free HD Medical Backgrounds WallpaperAccess
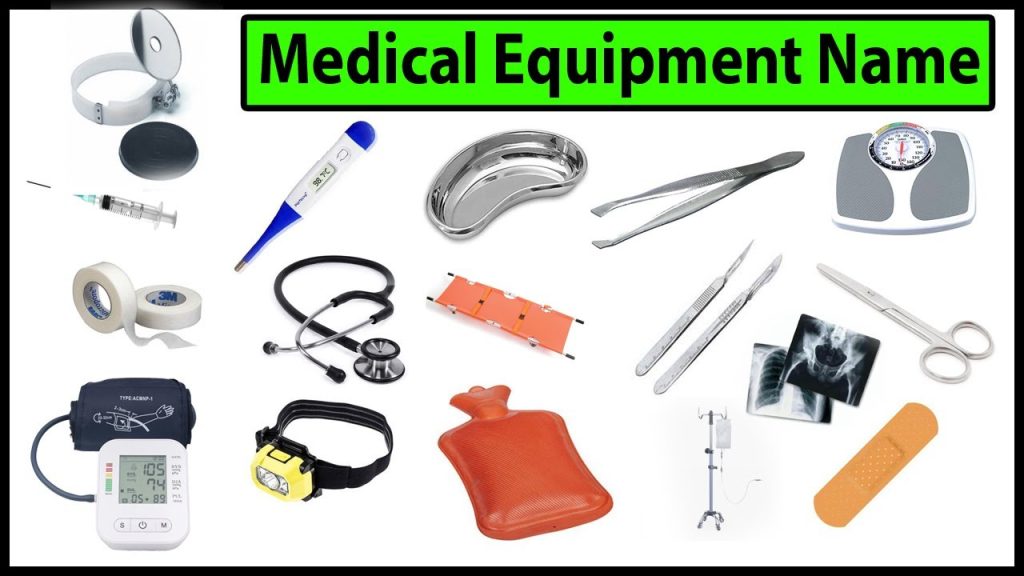
Basic Medical Equipment List Required in a Hospital Health Tips

All about the Pharmaceutical and Medical Device Industry Infographic

What Is A Medicine Tech at Kaitlyn Maund blog

Benefits of AI in Healthcare

What is the Most Important Medical Equipment?

Discover the Latest Advances in Medical Technology PaxeraHealth
_1662041110.png)
Decoding The Future Of Medical Technology Skill Lync Blogs

Medical Imaging Diagnostics at Bradley Briseno blog

Healthcare in India Sattva Consulting

Virtual Reality Training for Healthcare: The New Tool in Medical Education

What is Medical Terminology? Explanations Helpful Resources

Medical Items Vocabulary ll 130 Medical Items Name In English With
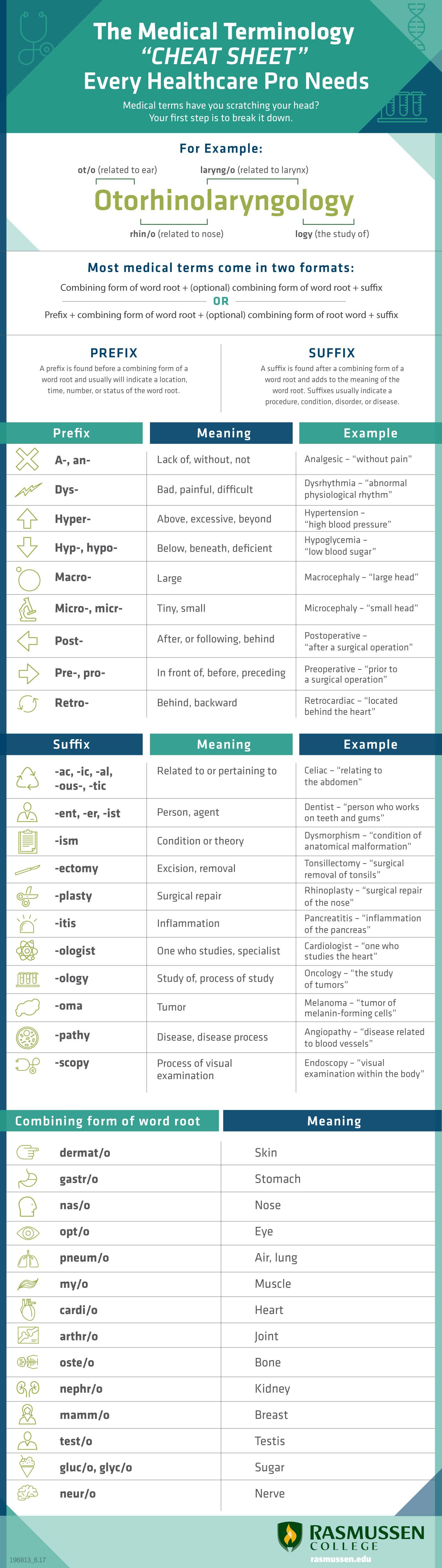
The Medical Terminology quot cheat sheet quot Every Healthcare Pro Needs

Medical Terminology: Learning Through Practice

15 Major Components of a Complete Medical Record

100 Healthcare Wallpapers Wallpapers com

List Of Basic Medical Terminology

Medical Doctor Symbol ClipArt Best

Medical Billing Coding Duties Job Description SCI

Simple Medical Terminology List

Medical Terminology List Printable Pdf Medical Prefixes and Suffixes

Medical Report What Is a Medical Report? Definition Types Uses
25 Inspiring Medical Website Design Examples (With Tips) Magezon

3d medical sign symbol concept icon 21658602 PNG

Common Medical Abbreviations and Terms You Should Know

Medical Doctor Symbol ClipArt Best
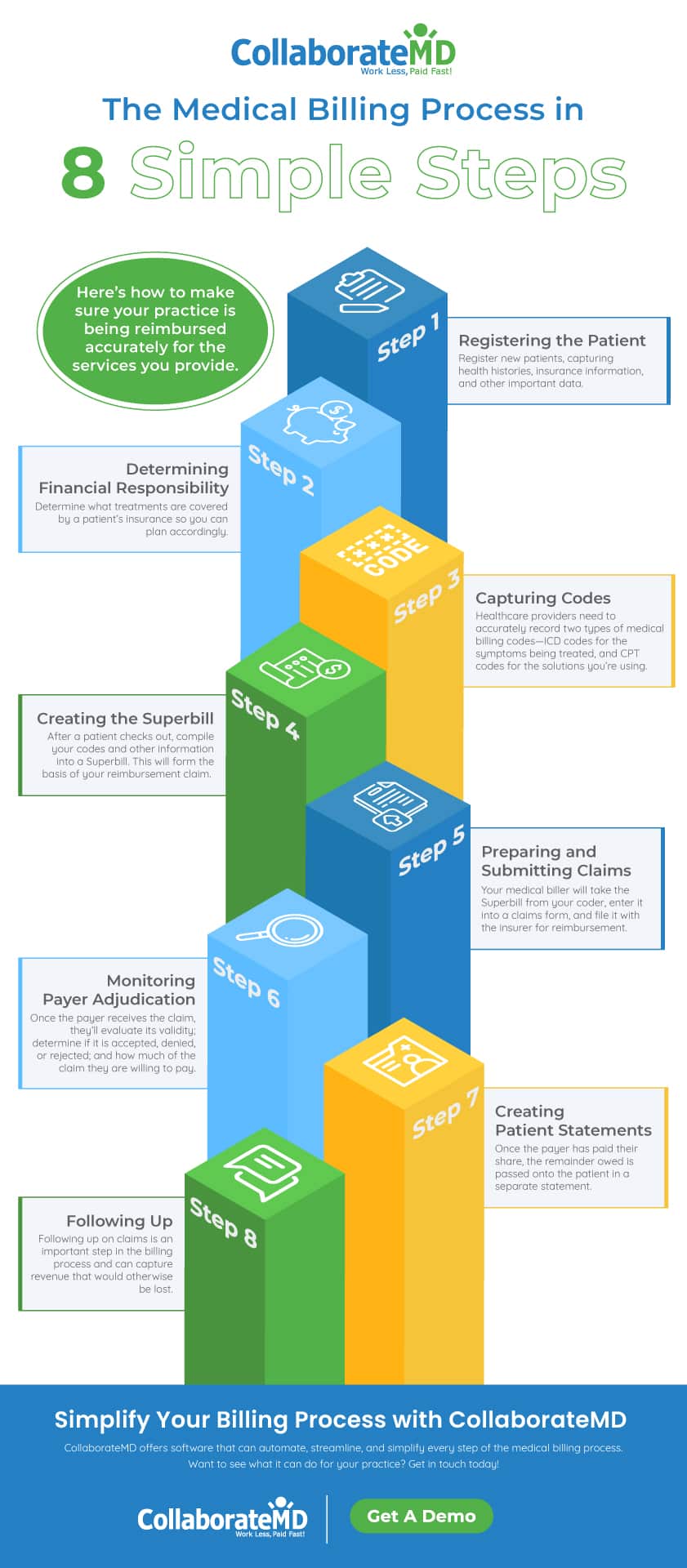
What Is the Medical Billing Process? Steps Cycle Overview
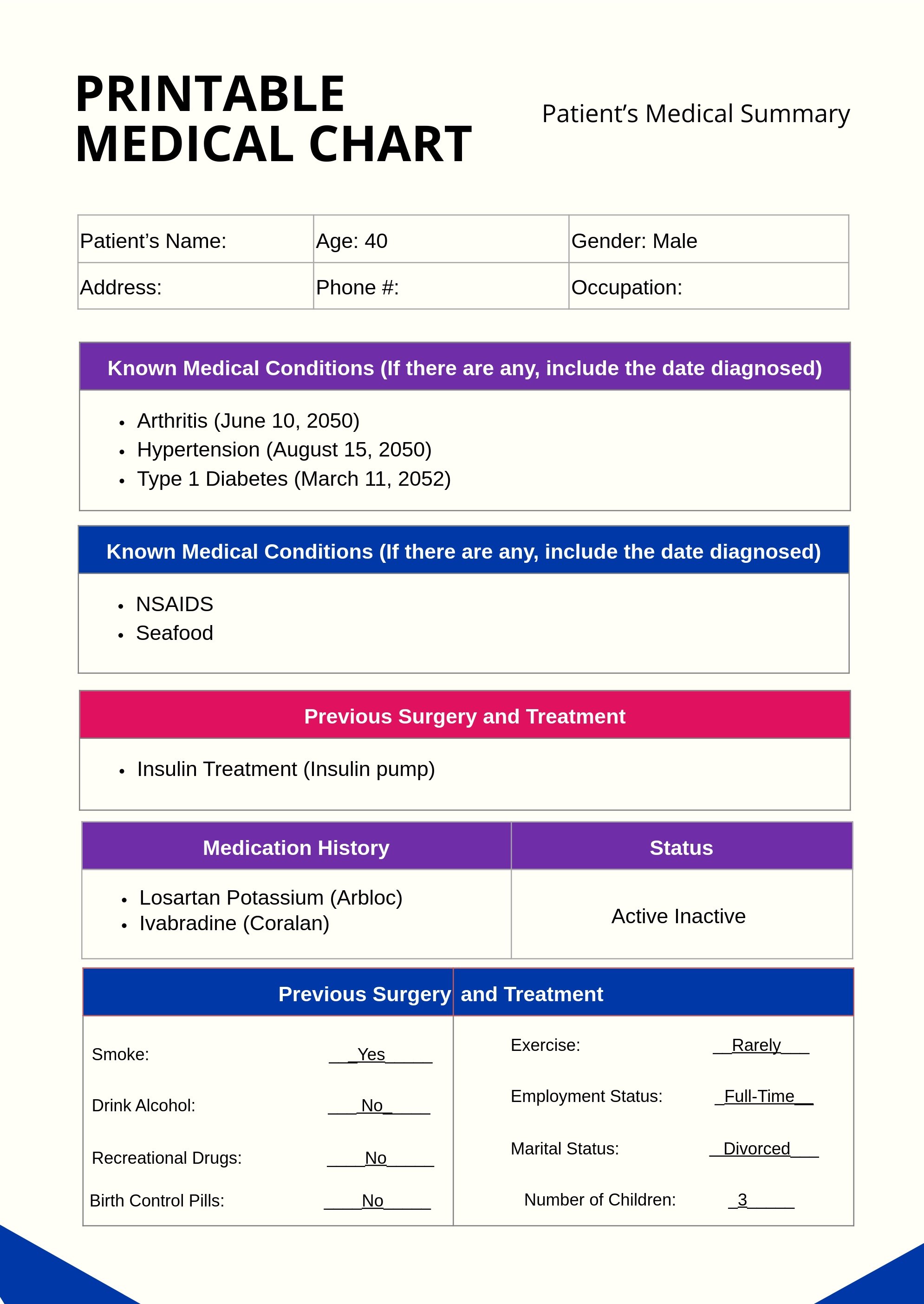
Free Medical Chart Templates Editable and Printable